US $1,999.99
| Condition | Used
:
An item that has been used previously. The item may have some signs of cosmetic wear, but is fully operational and functions as intended. This item may be a floor model or store return that has been used. See the seller’s listing for full details and description of any imperfections.
|
| Seller Notes | “Does Not Include Chuck Key...Has scuffs/scratches” |
Directions
Similar products from Orthopedic Surgical Instruments

DEPUY 7.0 MM CANNULATED SCREW FIXATION SYSTEM *

3 Trays EBI Orthofix Dynamic Axial Fixation Screw Drill Instrument Lot 240p-35lb

ZIMMER / HALL 5044 DRILL SET @

V.Mueller® U-0350 Kerrison Rongeur,Neuro Spine 90°UP 2mm12" Surgical Instruments
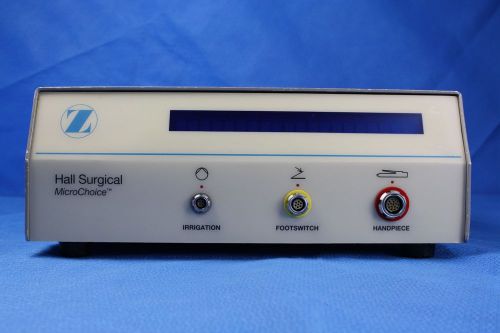
Hall Surgical MicroChoice 5020-020

12 Liston Bone Cutting Forceps 7.5" Surgical Instrument

Linvatec Microchoice MC 9840 Shaver
Jarit Bailey Gibbon Stainless Steel Rib Contractor 7" Cat # 300-235

Stryker Cutter Cannula with Holes & Trocar 270-713-300

Stryker CRANIO FIX TITANIUM CLAMP SYSTEM Spine Orthopedic Instruments

LINK AMERICA MODIFIED BANKART INSTRUMENTS W/CASE

***LOOK***Stryker System 5 Battery Charger - 4110-120

Capener Gouges for Orthopedic Spine Joint Surgery (NEW)
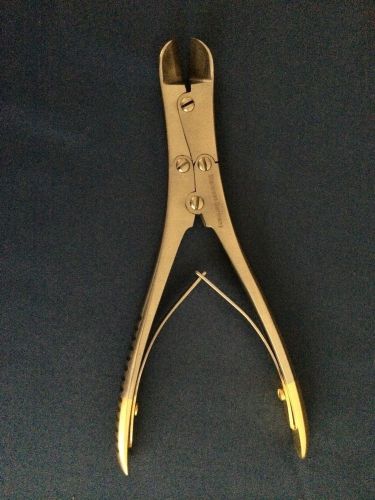
T/C MULLER CLAUS Pin+Wire Cutter 7" Lot of 2 Orthopedic Surgical Instruments

Parallel Wire Cutting Plier Surgical Dental Instruments

VINTAGE SYNTHES CARRYING CASE FOR ORTHOPEDIC TOOLS HARDWARE
People who viewed this item also vieved

Abaxis VetScan HM5 Veterinary Hematology Analyzer
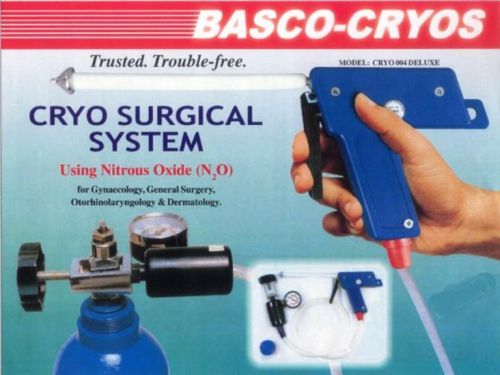
BASCO CRYO SURGICAL GUN USING NITROUS OXIDE SURGERY (N2O) Cryo Gun HYTHTR
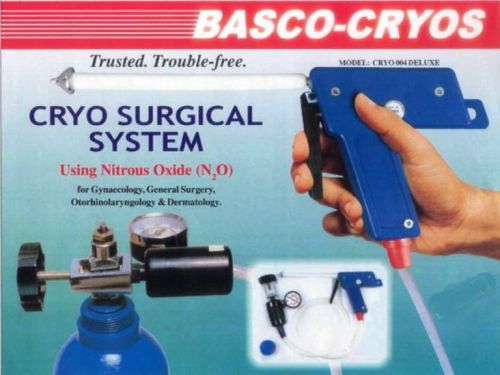
ENT CRYO SURGICAL SYSTEM OTORHINOLARY GYNAECOLOGY,WITH 5 PROBES Super Delux $

Electro Surgical Generator Model Simplex - 300 Monopolar Machine YTRU873456@!

Stryker Gaymar 38 cm x 55 cm TP22E Mul-T-Pad Temperature Pad Box of 20

CHATTANOOGA INTELECT VMS ELECTRICAL MUSCLE STIMULATOR

Back2Life Continuous Passive Motion

power supply puritan bennett 840

Drager 2 Each 4110906-02 5VDC Solenoid Valve for Narkomed Anesthesia New CS-823

puritan bennett , blacklight board

"The Triad Workshop Series Orientation Classes" 3 videos = 3 Health talks

LARGE ERCHONIA Cold Laser Great Condition

Pill Organizer Box wd Snap Lids 7dayAM/PM Large Comprtments for Bigger Pils IK11

Stainless Steel Enema Kit with PVC Tubing: 2 Quart Container. No Latex IK10

Multi-purpose Anti Pollution Mask Protection From Odors, Gases and Dust IK9
By clicking "Accept All Cookies", you agree to the storing of cookies on your device to enhance site navigation, analyze site usage, and assist in our marketing efforts.
Accept All Cookies